The Department of Science & Technology (DST) has funded ‘Module Innovations”, a Pune-based healthcare startup working on point of care diagnostics to develop its platform technology for rapid diagnosis of diseases to develop a test kit for detecting COVID-19 within 10 to 15 minutes.
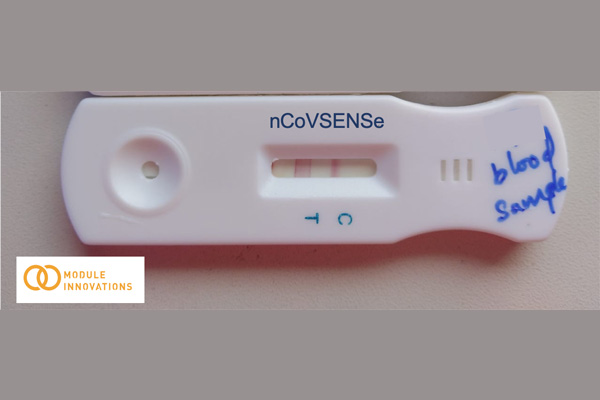
Using the proven concept from its flagship product ‘USense’, Module is now developing nCoVSENSEs (TM) which is a rapid test device for detection of antibodies that have been generated against the COVID-19 in the human body.
As per the current stage that India is in, doing a mass screening for the viral infection is of utmost importance. The rapid test device will enable the healthcare personnel to confirm infection in patients. Also, the kit will help in determining whether an infected patient has recovered and if not then at what stage of infection in the patients.
The presently used testing confirmatory method of Real-Time Reverse Transcription Polymerase Chain Reaction (RT-PCR) though a gold standard is expensive and takes a longer time. Moreover, trained manpower is required to carry on the tests. The newly developed rapid test will help manage the problem in a more efficient way at a lesser cost.
According to Professor Ashutosh, Secretary, DST, “While not being a substitute for the PCR based confirmatory technique, the tests based on the detection of antibodies are being deployed globally for the purposes of rapid mass screenings that take some of the burdens off from a limited number of PCR machines, and help in the formulation of strategies and decision making, among other things.”
The nCoVSENSe test aims to detect the IgG and IgM antibodies that are generated in the human body upon the onset of viral infection and is targeted against the spike proteins making it specific for COVID-19 infection.
Also Read: Govt of India launches ‘Aarogya Setu’ app to track COVID19 infected
The startup is planning to deploy the test in a time of 2-3 months after due validation from the national agencies. The test kit in future will also help determine people who have recovered and assign them front line jobs. The test could also be used to screen patients and passengers at airports, railway stations, hospitals and many such settings thus safeguarding us for any future outbreak as well.
While the feasibility of the technology has been proved, the Proof of concept (PoC) and Prototype illustrating functionality of the product are to be demonstrated.
Also Read: JNCASR develops antimicrobial coat against COVID-19 to prevent transmission
Be a part of Elets Collaborative Initiatives. Join Us for Upcoming Events and explore business opportunities. Like us on Facebook , connect with us on LinkedIn and follow us on Twitter, Instagram.
"Exciting news! Elets technomedia is now on WhatsApp Channels Subscribe today by clicking the link and stay updated with the latest insights!" Click here!